Over a decade, CAR T cell therapy has evolved from a bold experimental concept into a clinical reality, reshaping oncology through remarkable clinical outcomes, particularly in hematological malignancies. Despite great success, the evolution of CAR-based therapies is far from over.
The field continues to innovate to address various challenges, including deficiencies in CAR T cell persistence in blood malignancies and limited efficacy in the solid tumor setting. To this end, significant effort has been devoted to developing CAR constructs with improved performance. For example, modifications to the CAR intracellular components, such as the insertion of co-stimulatory domains, aim to enhance T cell activation, function, and persistence. Additionally, engineering new functionalities by including cytokine-induction domains (e.g., IL-12 inducers) and regulatory switches (e.g., small-molecule switches) aims to improve the efficacy and safety of cell therapies [1,2].
Advances are not limited to intracellular domain components; the field also continues to evolve CAR extracellular binding domains (ECD) for improved performance. Optimizing ECD affinity and stability has emerged as a critical step towards generating cell therapies with improved discrimination and persistence. Moreover, immunogenicity concerns are driving developers to leverage fully human compact binders, such as single VH domains derived from HCAb humanized transgenic or phage display platforms, in the development of CAR therapies.
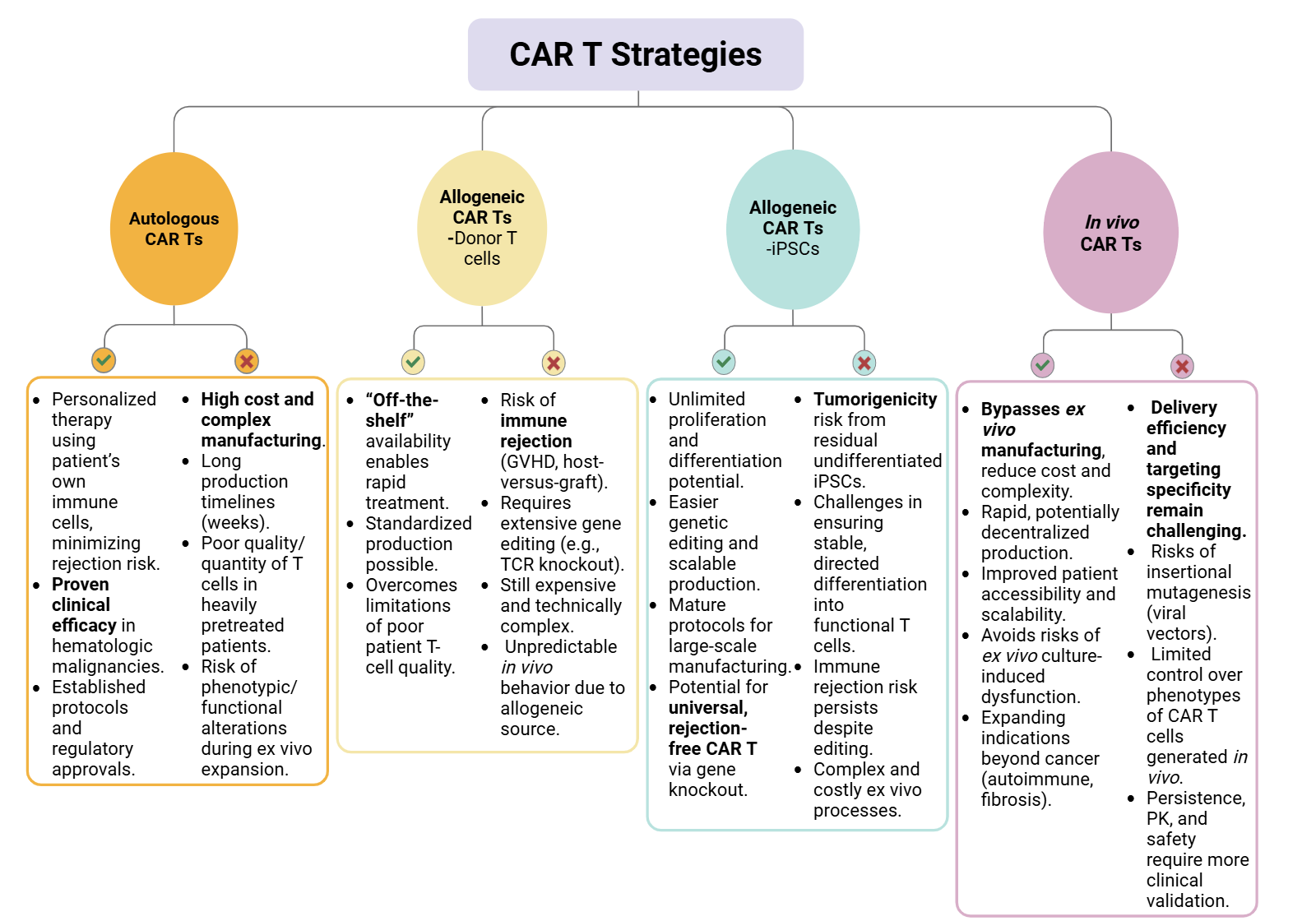
Amid all the progress, a major challenge of all CAR cell therapies approved to date and many in the clinical pipeline is their highly personalized nature. Developing these autologous CAR T cell therapies presents a series of challenges in cell sourcing, manufacturing standardization, and timely patient delivery. Furthermore, the complexities of ex vivo manufacturing have driven up the cost of these potentially life-saving therapies, making them inaccessible to many patients.
Autologous ex vivo CAR T cell therapy workflow

These challenges have pushed the field to innovate alternative solutions. For example, allogeneic (i.e., donor-derived) and universal (i.e., iPSC) CAR cell therapies can improve access by eliminating the challenges of sourcing T cells from patients with relapsed/refractory malignancies. Additionally, these strategies can help expedite manufacturing, reducing lag time to infusion. However, manufacturing cell therapies from donor T cells requires the implementation of gene-editing strategies to prevent immune rejection. Similarly, while the potential to develop a universal source of rejection-free iPSCs is a significant advantage, differentiation and validation of the resulting T cell product adds a layer of complexity [3].
Ultimately, both strategies rely on challenging, lengthy ex vivo manufacturing processes and do not overcome the cost barrier. Therefore, in the evolving landscape of CAR-based therapies, in vivo CAR T cell production is emerging as a promising solution to many of the current challenges.
Comparison of CAR T cell strategies
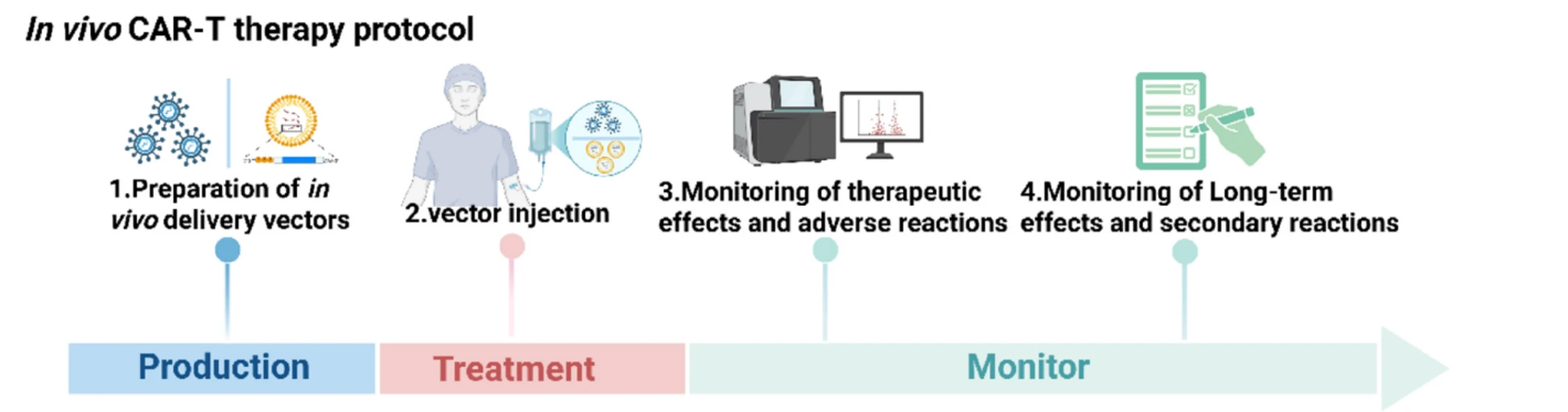
In vivo CAR: How does it work?
The proposal seems simple enough- deliver CAR constructs into patients to produce cell therapies within the body. Such an approach would essentially eliminate treatment lag, solve most manufacturing complexities, and significantly reduce cost. Nevertheless, as with any gene-based therapy, efficient and precise targeted delivery represents a significant challenge.
In vivo CAR T cell therapy workflow
Currently, developers are leveraging various delivery systems to target T cells, including viral vectors, nanoparticles, and implantable biocompatible scaffolds.
Viral vectors
The success of lentiviral vectors in developing CAR T cells ex vivo has made them a preferred choice for in vivo CAR delivery. However, lentivirus vectors do not enable T cell targeting, carry a risk of insertional mutagenesis, and can induce adverse immune reactions [3].
An emerging strategy in the field to target T cells specifically is the use of engineered lentiviral vectors that express CD8- and CD4-targeting molecules (i.e., CD8-LV and CD4-LV). Preclinical studies have shown that targeted lentiviral vectors can enable selective and efficient delivery of CAR to specific T cell subsets, resulting in the in vivo generation of CD19-CAR T cells that effectively kill tumor cells [4,5].
Several biotech companies are leading the way in implementing lentiviral CAR delivery at the clinical stage, including Umoja Biopharma, Kelonia Therapeutics, and Interius Biotherapeutics. Among these, Umoja’s UB-V111, enabled by their VivoVec platform, was the first in vivo CAR T cell therapy to receive Investigational New Drug (IND) application clearance by the FDA [6,7].
In vivo CAR candidates undergoing clinical evaluation
| Company | Vector/Platform | Lead Target | Indication | Clinical Status |
|---|---|---|---|---|
| Umoja Biopharma | Lentiviral vector / VivoVec | CD19 (UB-VV111) | B cell malignancies | Phase 1, Fast Track |
| Interius BioTherapeutics | Lentiviral vector | CD20 (INT2104) | B cell malignancies | Phase 1 |
| Kelonia Therapeutics | Lentiviral vector / iGPS | BCMA (KLN-1010) | Multiple Myeloma | Phase 1 |
In vivo CAR candidates undergoing clinical evaluation from top developers: Umoja, Interius, and Kelonia
Nanoparticles
The COVID-19 pandemic was a pivotal moment for lipid nanoparticles (LNPs), transforming them into a widely accepted technology that played a crucial role in the rapid development of mRNA vaccines. LNPs have demonstrated their ability to stabilize RNA, enhance cellular uptake, and provide scalability, establishing them as a fundamental component of modern drug delivery and a driving force behind the increasing prominence of RNA therapeutics.
Leveraging LNPs for in vivo CAR delivery has several advantages. LNPs have a confirmed favorable safety profile, and their production is scalable and less costly than viral production. Delivering CAR constructs as mRNA eliminates the risks of insertional mutagenesis associated with viral vectors and supports transient rather than long-term CAR expression [3]. This is particularly important as CAR-based cell therapy applications expand beyond cancer to include autoimmune and infectious diseases. In those contexts, transient CAR expression may enhance safety and improve treatment control [8,9].
Despite the benefits of LNPs, similar to viral vectors, targeting specificity remains challenging. Surface modification of lipid nanoparticles (LNPs) with antibodies, antibody fragments, and, more recently, compact binders, such as nanobodies (VHH domains) or fully human VH single domains, has emerged as a strategy for targeting various T cell receptors. This includes T cell subset markers such as CD4 and CD8, as well as pan-T cell markers such as CD3 and CD7. This approach has been validated in vitro and is applicable for in vivo T cell targeting [9,10,11].
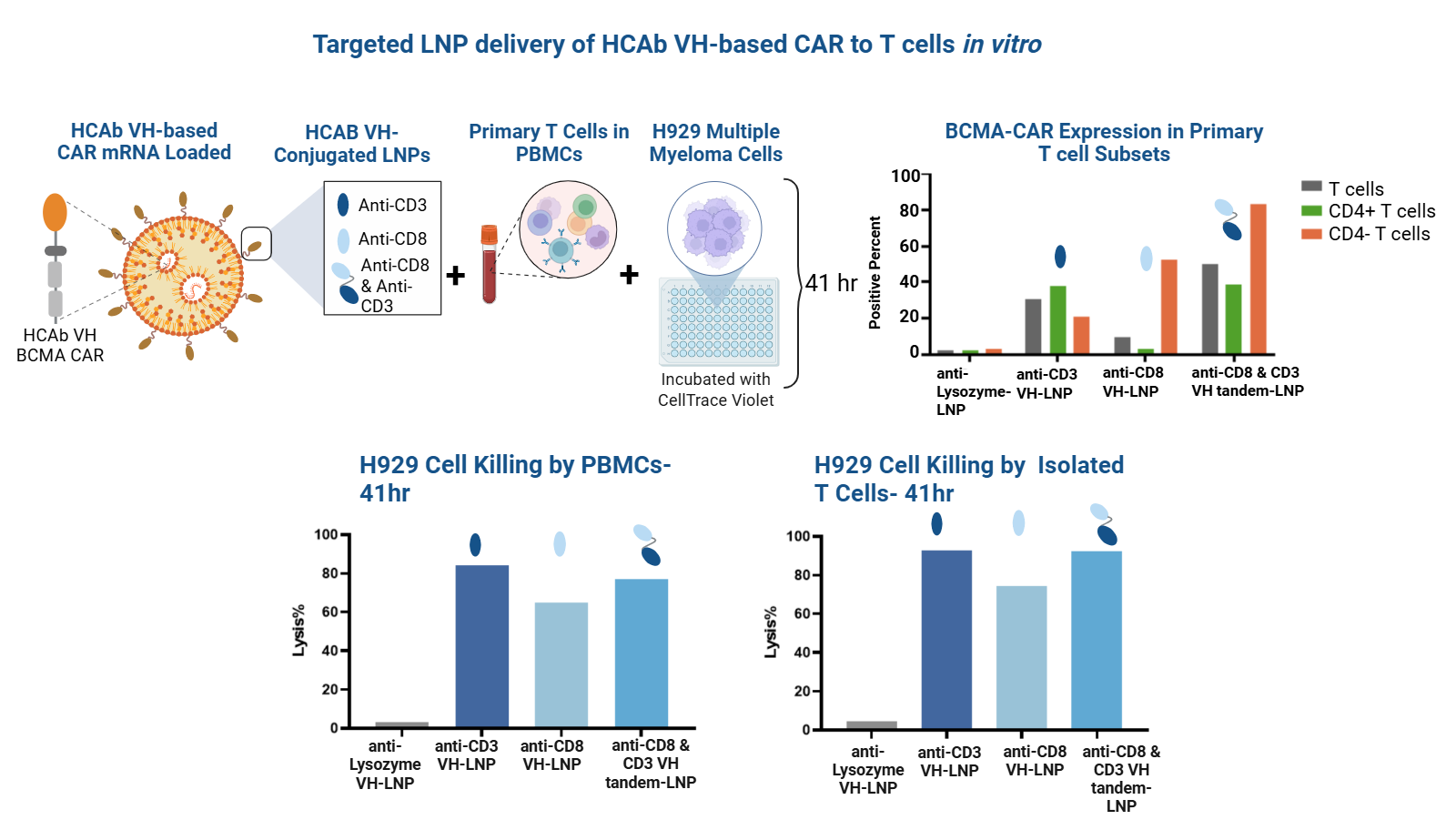
In vivo CAR delivery via functionalized LNPs has progressed rapidly from concept and preclinical studies to clinical trials. Recently, a small group of patients with systemic lupus erythematosus received CD19-CAR mRNA LNPs functionalized with anti-CD8 VHHs. Significantly, complete B-cell depletion was achieved in some patients with this approach, and no high-grade cytokine release syndrome or neurotoxicity was observed [12].
Functionalization of LNPs for CAR delivery to T cells
Nanoparticle-enabled in vivo CAR programs
| CAR Specificity/Sequence | CAR Indication | Nanoparticle Type | Conjugated Binder Target | Binder Format | Stage | Link |
|---|---|---|---|---|---|---|
| FAP/mRNA | Cardiac disease | LNP | CD5 | IgG | Preclinical | 2022 |
| CD19/mRNA | Autoimmune disease | LNP | CD3, CD7 | Fab | Preclinical | 2023 |
| CD19/mcDNA-CAR and mRNA-transposase | Cancer | LNP | CD7, CD3 | VHH, scFv | Preclinical | 2025 |
| CD19/mRNA | Autoimmune disease | LNP | CD8 | VHH | Preclinical and Clinical | 2025 |
Preclinical and clinical strategies for the functionalization of LNPs for CAR delivery to T cells
First-in-human studies are also underway for GT801 from Vivacta Bio, a T-LNP/mRNA–based in vivo CAR-T product candidate targeting CD19. Specific delivery of GT801 is enabled by Grit Bio’s proprietary CLAMP (Controllable Ligand Attachment Modification and Purification) technology for site-specific antibody conjugation [13,14]. Initial findings reported at the 2025 American Society of Hematology (ASH) Annual Meeting indicate GT801-positive outcomes in both efficacy and safety in patients with non-Hodgkin lymphoma. Significantly, the Phase 1 study is demonstrating the versatility of the approach by enabling repeated dosing, inducing robust CAR expression selectively in T cells, and supporting substantial B cell depletion [15].
Beyond delivering to T cells, in vivo CAR strategies are being leveraged to target myeloid cells. For instance, CREATE Medicines’ MT-303 is an experimental, in vivo GPC3-targeted CAR therapy that selectively programs myeloid cells. Phase 1/2 studies are demonstrating CAR expression in both circulating and tumor-infiltrating myeloid cells, myeloid activation and GPC3-positive tumor cell cytotoxicity, and immune-modulating effects [16].
Implantable scaffolds
Amongst in vivo CAR approaches, the use of implantable scaffold biomaterials remains unexplored at the clinical stage. Nevertheless, the concept is being actively evaluated in preclinical and translational studies.
One strategy leveraging biomaterials involves isolating T cells from patients to seed implantable scaffolds. For instance, in the multifunctional alginate scaffold for T cell engineering and release (MASTER) approach, T cells and CAR-encoding viral particles are packaged ex vivo and can be rapidly implanted in the patient without delay [17]. Once implanted, the scaffold serves as a homing center for T cell transduction. Additionally, key molecules conjugated to the scaffold promote T cell activation and expansion, including antibodies targeting CD3 and CD8, as well as IL-2.
Similarly, a second alignate-based scaffold, Drydux, follows a similar working principle. Preclinical in vivo studies have demonstrated that both scaffolds can support the generation of functional CAR-T cells with improved persistence and efficient tumor growth control [3,17,18].
Another strategy leveraging biomaterial-based implants by-passes the need for T cell isolation. By functionalizing scaffolds (e.g., porous collagen scaffolds) with CAR-encoding vectors, T cell chemoattractant and stimulatory factors, these implants enable in vivo generation of CAR T cells [3,19].
Empowering in vivo CAR strategies through modular VH-domains
Advancing in vivo CAR therapies requires platforms that seamlessly integrate highquality binder discovery with formats compatible for direct inbody expression. Fully human HCAb VH domains offer an ideal foundation for this shift, enabling compact, stable, and highly expressible CAR architectures suited for viral, nonviral, and mRNA–LNP delivery.
Nona Biosciences accelerates this transition through its NonaCAR™ and NonaCARFx™ platforms, which pair fully-human HCAb VH-domain discovery with functional CAR-format screening to identify binders with optimal biophysical properties, reduced immunogenicity, and able to support robust signaling.
Beyond supporting CAR binding, HCAb VH domains can be directly used to functionalize LNPs and lentiviral particles, two major delivery modalities leading in vivo CAR strategies.
Together, these technologies create a unified workflow empowering researchers to design next-generation in vivo CAR-T, CAR-NK, and CAR-M therapies with greater precision, scalability, and translational potential.
Related Resources
To explore CAR extracellular domain design and functional screening approaches in more detail:
- Download our Poster on Fully Human VH CAR platforms for a concise, a concise, visual overview of HCAb-derived VH domains applied in CAR-format screening, functional validation, and targeted delivery workflows
- Read our Related Blog on Functional Screening for a focused discussion on how ECD properties are assessed and optimized for solid tumor applications
- Bui T. A. et al., eBioMedicine, 2024. [Link]
- Lu L. et al., Sci Adv, 2024. [Link]
- Chen Y. et al., Experimental Hematology & Oncology, 2025. [Link]
- Pfeiffer A. et al., EMBO Mol Med, 2018. [Link]
- Agarwal S. et al., Molecular Therapy, 2020. [Link]
- Umoja Biopharma, Umoja Biopharma Announces that UB-VV111 Receives FDA Fast Track Designation for Relapsed/Refractory B-Cell Malignancies, 2025. [Link]
- Michels K. R. et al., J Immunother Cancer, 2023. [Link]
- Kitte R. et al., International Journal of Molecular Sciences, 2025. [Link]
- Billingsley M. M. et al., Small, 2023. [Link]
- Fernandez Bimbo J. et al., J Immunother Cancer, 2025. [Link]
- Tombacz I. et al., Molecular Therapy, 2021. [Link]
- Wang Q. et al., NEJM, 2025. [Link]
- Vivacta Bio, Vivacta Bio Announces Promising First-in-Human Results for GT801, an In Vivo CAR-T Therapy, in Non-Hodgkin’s Lymphoma at the 2025 ASH Annual Meeting, 2025. [Link]
- Sun J. et al., J Immunother Cancer, 2025. [Link]
- Vivacta Bio, Vivacta Bio Unveils Groundbreaking Results for GT801 In Vivo CAR-T Therapy at 2025 ASH Annual Meeting, 2025. [Link]
- CREATE Medicines, CREATE Medicines Doses First Patient in Frontline HCC Trial Evaluating MT-303, an In Vivo CAR Therapy, in Combination with Standard-of-Care Immunotherapy, 2025. [Link]
- Agarwalla P. et al., Nat Biotechnol, 2025. [Link]
- Pandit S. et al., Biomaterials, 2025. [Link]
- Inamdar V. V. et al., J Controlled Release, 2024. [Link]