The Rise of CAR-T Cell Therapies
Cancer treatment has traditionally relied on chemotherapy, radiation, and surgery. However, in the past 20 years, cancer immunotherapy has been transforming the field of oncology through molecular pathway engineering to redirect the immune system to recognize and destroy malignant cells. The concept of genetically reprogramming T cells to target specific antigens was first described in 1987 by Yoshihisa Kuwana et al. at Fujita Health University. By fusing immunoglobulin-derived variable regions, specific for phosphorylcholine, with T-cell receptor (TCR) constant regions, Kuwana and colleagues enabled T cells to respond to the antigen independently of MHC presentation [1]. Separately, in 1989, Gross, Waks, and Eshhar at the Weizmann Institute, went on to similarly modify a T cell receptor to recognize the hapten, 2,4,6-trinitrophenyl (TNP) [2]. Transfection of the chimeric antigen receptor into a cytotoxic T-cell hybridoma enabled the expression of a functional receptor with the capacity for MHC-unrestricted TNP binding. The innovation of swapping the TCR variable region with that of an antibody-derived binding domain enabled the creation of the first chimeric antigen receptor (CAR). As a result, the genetically modified T cells were able to identify targets specifically and induce cellular cytotoxicity.
Decades of optimization in antibody design and development, humanization, gene-transfer technologies, T-cell manufacturing, dosing strategies, and more followed these early proofs of concept.
A significant milestone for the clinical translation of the CAR concept was achieved by Carl June, who demonstrated that genetically modified T cells could persist in HIV patients, revealing their therapeutic potential [3]. He then adapted this approach to cancer, engineering patient-derived T cells with chimeric antigen receptors to target malignancies, making the first CAR-T cell therapy a reality [4].
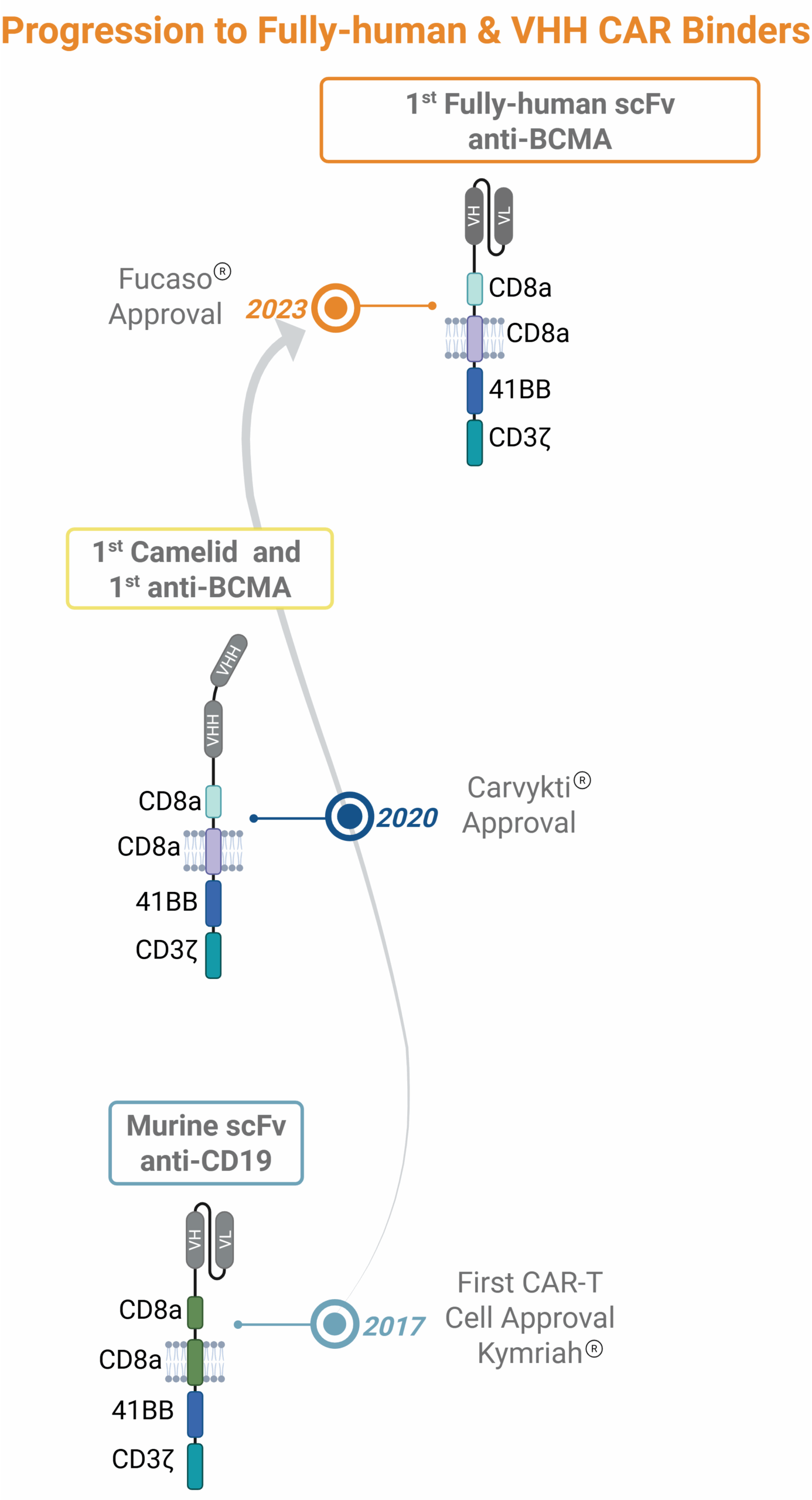
The field reached a major milestone in 2017 with the FDA approval of tisagenlecleucel (Kymriah®). This breakthrough introduced a revolutionary approach to treating B-cell Acute Lymphoblastic Leukemia (B-ALL) and B-cell lymphomas. By equipping T cells with tumor-specific CARs, researchers achieved an effective strategy to enhance immune responses against cancer cells and induce durable responses [4]. Kymriah demonstrated significant efficacy in treating blood malignancies in children, while axicabtagene ciloleucel (Yescarta®), the second approval in 2020, showed similar success in adult patients. These first approvals paved the way for implementing CAR-based cell therapies in cancer and beyond.
Since these first approvals, CAR-T cell therapy has expanded rapidly. To date, eleven autologous CAR-T cell therapies have received first approval by the FDA (7) and NMPA (4), all indicated for relapsed or refractory hematological malignancies. Most of these therapies target the cancer antigen CD19, although more recent approvals include CAR-T cells specifically designed to target BCMA-expressing cancer cells [5].
| Brand Name | Generic Name | Target | Antigen Binder | First Approval | Indication |
|---|---|---|---|---|---|
| Kymriah | Tisagenlecleucel | CD19 | scFv, murine | 2017 / FDA | B-cell acute lymphoblastic leukemia (B-ALL) |
| Yescarta | Axicabtagene ciloleucel | CD19 | scFv, murine | 2017 / FDA | Large B-cell lymphoma |
| Tecartus | Brexucabtagene autoleucel | CD19 | scFv, murine | 2020 / FDA | B-cell precursor acute lymphoblastic leukemia |
| Breyanzi | Lisocabtagene maraleucel | CD19 | scFv, murine | 2021 / FDA | Large B-cell lymphoma |
| Carteyva | Relma cel | CD19 | scFv, murine | 2021 / NMPA | Large B-cell lymphoma |
| Abecma | Idecabtagene vicleucel | BCMA | scFv, murine | 2021 / FDA | Multiple myeloma |
| Carvykti | Ciltacabtagene autoleucel | BCMA | VHHs, camelid | 2022 / FDA | Multiple myeloma |
| Inati-cel | Intaticabtagene autoleucel | CD19 | scFv, murine (HI19a hybridoma) | 2023 / NMPA | B-cell acute lymphoblastic leukemia (B-ALL) |
| Fucaso | Equecabtagene autoleucel | BCMA | Fully human scFv | 2023 / NMPA | Multiple myeloma |
| Aucatzyl | Obecabtagene autoleucel | CD19 | scFv, murine | 2024 / FDA | B-cell acute lymphoblastic leukemia (B-ALL) |
| Zervo-cel | Zevorcabtagene autoleucel | BCMA | Fully human scFv | 2024 / NMPA | Multiple myeloma |
More astounding is the number of registered CAR-T cell clinical studies. By mid-2024, CAR-T cell interventional clinical trials had amassed a total of over 1,400 studies globally. Significantly, despite the robust growth, most of these studies remained at early clinical stages [8]. Currently, a simple search on ClinicaTrials.gov for interventional CAR-T cell therapies reports over 1,700 studies, with a majority in Phase 1 evaluation.
Beyond Blood Cancers and Challenges Ahead
Building on the success of CAR-T cell therapies in blood cancers, the field continues to expand on their application to address solid tumors, autoimmune diseases, and infectious diseases [6]. Amongst potential new indications, solid tumors have proved to be a significant challenge for CAR-T cell therapies. Different factors converge in solid tumors, making them especially inaccessible to CAR-T cell therapy.
Critically, solid tumors present both physical and molecular barriers that act to limit CAR-T cell infiltration, function, and survival [8]. The highly heterogeneous cellular composition of solid tumors figures prominently as a barrier to CAR-T cell therapy success. The higher cellular and molecular complexity makes it extremely challenging to select unique tumor-specific antigens (TSA) to achieve efficient and safe targeting with CAR-T cells. Instead, tumor-associated antigens (TAAs), which are overexpressed in the tumor and present only at low levels in normal tissues, are often selected as therapeutic targets.
However, despite the low expression of tumor-associated antigens (TAAs) in normal tissues, targeting them with CAR-T cells often results in on-target off-tumor toxicities, limiting therapeutic outcomes [5].
Solid tumor heterogeneity is also tightly interlinked with the development of resistance to CAR-T cell therapy. Resistance may arise through antigen-escape mechanisms, such as the silencing or downregulation of antigen expression [10,11].
It’s important to recognize that CAR-T cell resistance can arise in various contexts, not just in solid tumors. In fact, disease relapse due to antigen escape is commonly seen in hematological malignancies following treatment with CAR-T cell therapies against CD19, BCMA, and other targets [8,10].
However, an important factor that adds complexity to solid tumors is their highly immunosuppressive microenvironment. This immunosuppression is driven by a variety of factors, including the unusually stiff and intricate extracellular matrix, which facilitates the presence of immunosuppressive cells such as regulatory T cells (T regs) and cancer-associated fibroblasts. The solid tumor microenvironment (TME) is also often hypoxic due to poor vascularization and saturated with immunosuppressive cytokines (e.g., TGF-β). Together, these factors not only limit CAR-T cell infiltration but also reduce their activity and persistence within the TME [5].
Innovative Approaches Overcome CAR-T Cell Therapy Challenges
To overcome the current challenges to therapeutic success, the field has continued to optimize CAR-modified T cells on several fronts. For instance, the evolution of the CAR molecule is far from over. Investigators continue to fine-tune and improve the composition, structure, and design of CARs, making them more responsive to low antigen levels.
From the original CAR designs, which leveraged a murine scFv domain, the field is moving towards the use of more compact VHH domains, which can be sourced from fully human heavy chain only antibodies (HCAbs), and overall, fully human binders. VHH binders simplify the path to produce multispecific modalities, making it easier to address the complex biology of solid tumors. Furthermore, the need for humanized and fully human CAR binders is highlighted by the documented anti-CAR immune responses observed in patients treated with CD19 CAR-T cells that utilize murine-derived scFv binders [12 ].
Pros and Cons
HCAb-derived VHH domains vs. scFvs as CAR Binders
| Feature | VHH-only CARs | scFv CARs |
|---|---|---|
| Size & Simplicity | Smaller and structurally simpler; easier to clone and express | Larger due to VH–VL linker; more complex folding and expression |
| Stability | Often more thermodynamically stable; reduced aggregation risk | Prone to misfolding or aggregation, especially under stress |
| Manufacturing Efficiency | Streamlined vector design; lower burden on viral packaging systems | Bulkier constructs may reduce viral titers and transduction efficiency |
| Immunogenicity | Potentially lower due to reduced linker and domain complexity | Linker regions and VL domains may increase immunogenicity |
| Target Accessibility | May better access cryptic or sterically hindered epitopes | Larger footprint can limit access to recessed antigens |
| Binding Affinity | May require affinity optimization; lacks VL contribution | Typically higher baseline affinity due to VH–VL pairing |
| Clinical Maturity | Emerging platform; fewer clinical trials to date | Well-established with multiple FDA-approved therapies |
Similarly, advances in the composition of intracellular signaling domains have been rapidly implemented, with five generations of CAR molecules developed to date [4]. Modifications to signaling domains not only make CARs more sensitive in the context of low target levels but also are meant to improve CAR-T cell long-term persistence [10].
To reduce the risk of on-target off-tumor toxicities, investigators continue to advance immunopeptidomic approaches. For instance, new discovery platforms are making it possible to identify novel tumor-specific antigens, which in turn could enable the development of CARs with enhanced specificity, efficacy, and safety. This is more recently exemplified by the development of IRIS, a computational platform that leverages the high incidence of RNA alternative splicing events in cancer cells to identify novel tumor-specific antigens for CAR therapy [13,14].
The identification of neoantigens expands the reach of CAR-T cells to epitope peptides derived from novel intracellular proteins. An emerging modality in the field is the development of CAR molecules with scFv or VHH TCR-like binders specific to neoantigen peptides, commonly referred to as TCR-like CARs [5,11,15,16].
To address the heterogeneity of solid tumors, as well as immunosuppressive and resistance mechanisms, strategies to reach and modulate multiple targets are quickly progressing. The development of multispecific CAR-T cells is creating opportunities to simultaneously reach more tumor antigens and engage more immune effectors.
Multi-targeting may be achieved by the coexpression of individual CARs, each having distinct antigen specificities (e.g., bicistronic), or by designing receptors with multiple antigen-binding domains (e.g., tandem scFvs or VHH domains). Alternatively, CAR-T cells with unique specificities can be used, as exemplified by sequential treatment strategies [9]. Several CAR-T cell therapies targeting two antigens are currently undergoing clinical evaluation for hematological malignancies (e.g., BCMA/CD19, BCMA/FcRL5, CD19/CD22) and solid tumors (e.g., VGFR1/PD-L1, Mesothelin/Claudin18.2, GD2/CD70), with most at the Phase 2 stage and based on bispecific CAR formats [17].
Increasing Access to CAR-T Cell Therapies
Despite the rapid growth of CAR-T cell therapies, accessing these potentially curative treatments remains a significant challenge for patients. The high cost, which is currently estimated to exceed $500,000 per treatment, often prevents patients from affording these therapies [8]. Additionally, the complex ex vivo manufacturing processes involved pose a major barrier to the timely delivery of high-quality cell therapies to patients [5].
Lastly, all approved products to date are manufactured from patient-derived or autologous T cells. Using autologous T cells is beneficial in that it eliminates the risk of graft-versus-host disease (GvHD). Nevertheless, obtaining healthy T cells in sufficient quantities from patients with late-stage refractory disease is often a challenge [17].
To address these major hurdles, the field is pivoting away from autologous-based CAR-T cells, which are manufactured from a patient’s own T cells, and developing strategies to enable the use of allogeneic T cells from healthy donors to create an “off-the-shelf” modality. This approach would enable the manufacture of ready-made CAR-T cell therapies for a broader patient population [17].
Another emerging strategy has the potential to enable patients to make their own CAR-T cells. In vivo CAR-T cell expression relies on nanocarrier-targeted delivery of mRNA encoding the CAR construct to T cells for transient expression [17,18].
Future Outlook
Looking ahead, CAR-T cell therapies are poised for transformative advancements that can redefine the landscape of cancer treatment. As researchers continue to innovate, exploring next-generation CAR designs and combinatorial approaches, there is invigorating potential to enhance the efficacy of these therapies against solid tumors and beyond. The pursuit of overcoming immunosuppressive microenvironments, developing novel targeting strategies, and harnessing gene editing technologies will pave the way for a more robust and versatile armamentarium in cancer therapy.
Generative AI approaches are transforming and accelerating the path to improved CARs, facilitating multiple processes, such as nominating targets, optimizing the design of receptors, predicting receptor expression, activity, and more. Overall, AI platforms are helping guide the engineering of CARs to address the many challenges imposed by the solid tumor microenvironment [19].
Notably, the field is also expanding to leverage other immune cell types for developing CAR therapies (e.g., NK cells and macrophages). Significant strides have been made in the development of CAR-NK cells, which, as innate immune cells, have intrinsic anti-tumor properties while also providing a safer therapeutic modality (e.g., decreased risk of cytokine release syndrome and neurotoxicity). More importantly, CAR-NK cells can be used as “off-the-shelf” therapies, overcoming a significant barrier faced by currently approved CAR-Ts. Despite these benefits, CAR-NK immunotherapies have yet to receive approval for commercial use mainly due to deficiencies in their long-term persistence [20].
Overall, the cell immunotherapy field is undergoing a period of explosive growth, focused on expanding CAR modalities, optimizing receptors, discovering better and safer targets, and leveraging the power of combinatorial approaches.
By addressing the existing challenges and expanding the therapeutic scope of various CAR-modified immune cells, the field is moving closer to a future where personalized, effective treatment options are accessible for all cancer patients, marking a significant leap toward improved survival rates and quality of life.
How Nona Biosciences Supports the Field
Advances in binder engineering, including fully human heavy chain only antibody (HCAb) formats for compact, highly developable CAR designs, are enabling multispecific targeting and improved performance in complex tumor environments. Using our NonaCAR™ fully human binders and NonaCARFx™ functional screening platform, Nona Biosciences identifies binders optimized for CAR expression and activation, supporting the development of next-generation CAR-T programs with greater precision.
- Kuwana Y. et al., Biochemical and Biophysical Research Communications, 1987. [Link]
- Gross G. et al., Proc Natl Acad Sci USA, 1989. [Link]
- Tebas P. et al., N Engl J Med, 2014. [Link]
- Gill S, June CH., Immunol Rev, 2015. [Link]
- Khan SH. et al., Front Immunol, 2025. [Link]
- Lin H. et al., Biomed Pharmacother, 2024. [Link]
- Keam S.J., Mol Diagn Ther, 2023. [Link]
- Cao LY. et al., Front Immunol, 2025. [Link]
- Li YR. et al., Trends Pharmacol Sci, 2024. [Link]
- Lu Y, Zhao F., Mol Cancer, 2025. [Link]
- Larson RC, Maus MV., Nat Rev Cancer, 2021. [Link]
- Gauthier J. et al., Blood Adv, 2025. [Link]
- Pan Y. et al., Proc Natl Acad Sci USA, 2023. [Link]
- Benton A. et al., Cancer Cell, 2025. [Link]
- Zhu L. et al., J Immunol Res, 2020. [Link]
- Walseng E. et al., Sci Rep. [Link]
- Qin H. et al., J Hematol Oncol, 2025. [Link]
- Parayath NN. et al., Nat Commun, 2020. [Link]
- Mergen M., bioRxiv, 2024. [Link]
- Zhong Y., Liu J., Cell Death Discov, 2024. [Link]