Antibody–drug conjugates (ADCs) are reshaping the oncology landscape, offering a way to deliver highly potent therapies with unprecedented precision. Yet their promise comes with complexity. As more ADCs advance into the clinic, particularly for solid tumors, which now account for the majority of recent approvals, the field faces the challenge of designing drugs that must balance stability in circulation, efficient tumor delivery, and potent intracellular activity. Moreover, because only a small fraction of each dose ever reaches the tumor, every component of the ADC: the antibody, the linker, and especially the payload must be carefully selected and optimized.
The rapid evolution of payload and linker technologies reflects this challenge. Traditional microtubule and DNA-damaging agents are now joined by a new generation of mechanisms, from topoisomerase inhibitors driving strong bystander effects to emerging modalities such as immune agonists, RNA polymerase inhibitors, and BCL-XL blockers. In parallel, linker innovation is accelerating, with improved Val-Cit chemistries, site-specific conjugation strategies, and tumor microenvironment-responsive designs pushing the boundaries of what ADCs can safely and effectively deliver.
Together, these advances signal a field in transformation. As developers refine each molecular component to overcome long-standing barriers, such as heterogeneous antigen expression, off-target toxicity, and limited payload penetration, ADCs are moving beyond their early constraints toward broader therapeutic impact. The result is a rapidly expanding toolbox that is redefining how targeted therapies are conceived, engineered, and deployed in the fight against cancer.
This momentum is already visible in the clinical landscape. Recent approvals reveal a decisive shift in where ADC innovation is taking hold, with solid tumor programs now dominating the field. Among clinical approvals since 2019, ADCs targeting solid tumor antigens predominate (75%) over those indicated for myelomas and lymphomas. This trend has intensified, with most approved ADCs since 2021 targeting antigens in gastric, cervical, ovarian, breast, and lung cancers [1].
As the therapeutic focus expands, so too does the need for linker and payload technologies capable of meeting the unique challenges posed by solid tumors, heterogeneity, dense stroma, and variable antigen expression.
Linker and Payloads in Clinically Approved and Advanced Clinical Stage ADCs
The evolution of ADC design is reflected in the diversity of linker-payload combinations now in clinical use. Seventeen ADCs have been approved by the FDA since 2000, and their molecular architecture highlights the field’s reliance on a few dominant chemistries.
The most frequently used combinations include cleavable mc-Val-Cit-PAB linkers paired with MMAE (35%), followed by mc-Gly-Gly-Phe-Gly linkers paired with deruxtecan (12%), and acid-labile hydrazone linkers paired with calicheamicin (12%) [1]. These patterns underscore how linker stability, cleavage specificity, and payload potency have shaped the first generation of clinically successful ADCs, while also revealing opportunities for innovation as developers push beyond established designs.
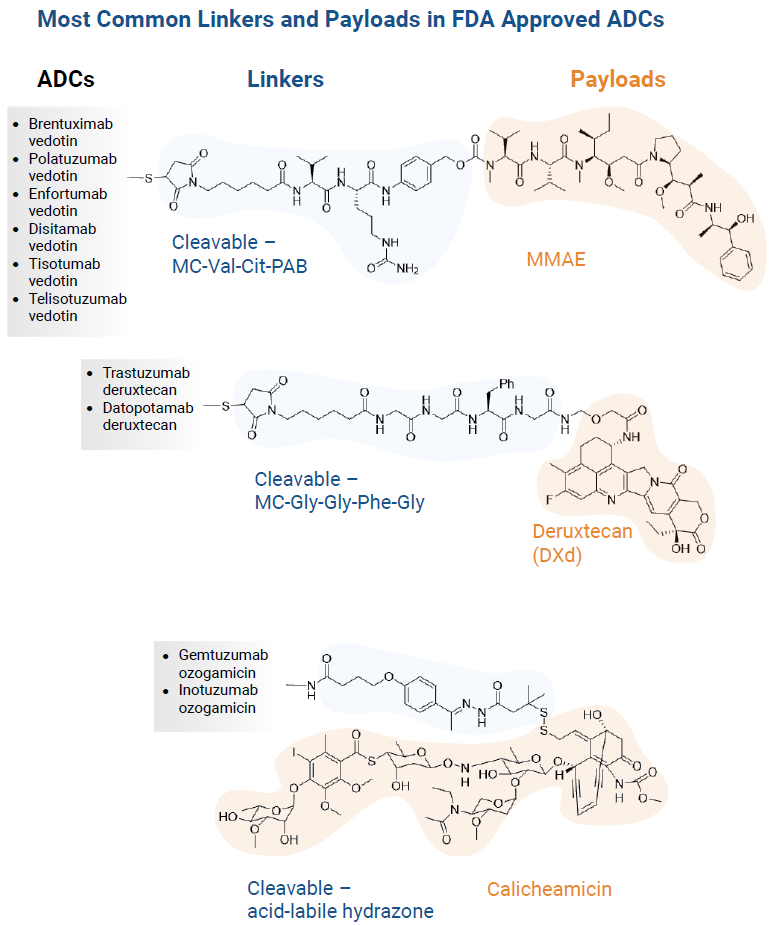
Ahead, we explore trends in the selection of linkers and payloads shaping ADCs at the advanced clinical stage, and emerging technologies designed to address remaining challenges in the efficacy and safety of these powerful drug modalities.
ADC Payloads
Payload selection remains one of the most consequential decisions in ADC engineering. Because only a small proportion of infused ADCs, roughly 2%, reach the tumor site, payloads must be exceptionally potent and stable under lysosomal conditions [2].
These cytotoxic agents act through mechanisms such as microtubule inhibition, DNA damage, and topoisomerase inhibition, each offering distinct advantages depending on tumor biology and therapeutic goals.
A notable shift is occurring in late-stage development as topoisomerase I (TOP1) inhibitors gain prominence. Among roughly 24 ADCs currently in Phase 3 trials, nearly half (~46%) use TOP1 inhibitor payloads, including adizutecan, DXd, exatecan, KL610023, P1003, SHR9265, and SN-38. This represents a significant increase compared to their use in FDA-approved ADCs (12%) [2].
The rise of TOP1 inhibitors is driven in part by their strong bystander effect, which enhances cytotoxic reach within heterogeneous solid tumors, precisely the settings where ADCs are now most actively being deployed.
ADCs in late-stage clinical studies using tubulin or TOPI inhibitory payloads
| ADC Candidates | Payload | MOA |
|---|---|---|
| Tusamitamab ravtansine (SAR408701) | DM4 | Microtubule inhibition |
| DP303c, MRG-002, MRG-003, Sigvotatug vedotin (SGN-B6A), Telisotuzumab vedotin, Zilovetramab vedotin | MMAE | |
| ARX-788, Depatuxizumab mafodotin (ABT-414), FS-1502 | MMAF | |
| Luveltamab tazevibulin (STRO-002) | SC209 | |
| Telisotuzumab adizutecan (ABBV-400) | Adizutecan | Topoisomerase I inhibition |
| Datopotamab deruxtecan, Ifinatamab deruxtecan, Patritumab deruxtecan, Raludotatug deruxtecan | DXd (deruxtecan) | |
| Rinatabart sesutecan | Exatecan | |
| Sacituzumab tirumotecan (MK-2870) | KL610023 | |
| BNT323 / DB-1303 | P1003 | |
| SHR-A1811 | SHR9265 | |
| FDA018 | SN-38 |
Information for the table above was obtained from Wang et al. (2025). [2]
Mechanisms of action of frequently used payloads
As payload trends shift, understanding how these agents exert their cytotoxic effects becomes increasingly important. Microtubule inhibitors and topoisomerase inhibitors remain foundational to ADC design, each offering distinct advantages depending on tumor biology and therapeutic goals.
Microtubule-inhibiting payloads such as MMAE, MMAF, DM1, and DM4 disrupt the structural framework required for cell division. By binding tubulin and preventing proper microtubule assembly, these agents induce mitotic arrest and ultimately cell death [3]. Their longstanding use reflects both their potency and their compatibility with established linker chemistries.
Topoisomerase I (TOP1) inhibitors, by contrast, act directly on DNA topology. Payloads such as deruxtecan stabilize the TOP1–DNA cleavage complex, preventing the relaxation of supercoiled DNA during transcription and replication. The resulting DNA damage and replication fork collapse trigger irreversible cytotoxicity [4]. Their growing prominence, particularly in Phase 3 clinical programs, stems from their strong bystander effect, which enhances tumor penetration and activity in heterogeneous solid tumors.
Mechanisms of action of less commonly used payloads
Beyond these dominant classes, several potent but less frequently used payload types contribute additional therapeutic mechanisms. DNA-damaging agents, including alkylators such as seco-DUBA, cross-linkers such as PBD dimers, and cleaving agents such as calicheamicin, offer exceptional potency but require careful control due to their broad cytotoxic potential [2]. SecoDUBA, for example, binds the minor groove of DNA and forms highly stable adducts that disrupt replication and transcription. Its membrane permeability also enables a strong bystander effect, a feature leveraged in trastuzumab duocarmazine (SYD985), now in Phase 3 evaluation [2,5,6].
Protein synthesis inhibitors represent another niche category. Payloads such as PE38 and ETA252608, derived from Pseudomonas aeruginosa exotoxin A, block peptide elongation by ADPribosylation of the eukaryotic elongation factor 2 (eEF2) [2,7]. Although rarely used, these agents demonstrate the breadth of mechanisms available to ADC developers.

Emerging ADC payloads
As the field pushes beyond traditional cytotoxics, a new generation of payload modalities is expanding what ADCs can achieve. These emerging mechanisms are designed not only to kill tumor cells directly but also to reshape the tumor microenvironment, overcome resistance, and broaden therapeutic windows.
Immune-activating payloads, such as STING and TLR agonists, are gaining traction for their ability to convert immunologically “cold” tumors into inflamed, immune-responsive environments [2, 9]. When delivered via immune-stimulating antibody conjugates (ISACs), these agents activate myeloid cells, enhance T-cell recruitment, and promote Fcγ receptor–mediated phagocytosis. Several candidates, including XMT2056, DS3610, and TAX500, are now in Phase 1 trials [10, 11, 12, 13].
RNA-inhibitory payloads, including α-amanitin and triptolide, offer a distinct advantage: the ability to kill both dividing and dormant tumor cells [2, 14]. This makes them particularly attractive for overcoming resistance mechanisms. To date, at least two antibody conjugates leveraging RNA polymerase II inhibitor payloads, HDP101 and KH815, have already entered clinical evaluation, with KH815 representing a first-in-class dual-payload design combining exatecan and triptolide [14, 15, 16].
BCL-XL inhibitors represent another emerging class aimed at overcoming apoptosis resistance. While systemic BCL-XL inhibitors have been limited by thrombocytopenia, neutropenia, and cardiovascular toxicity, antibody-mediated delivery offers a path to safer, more targeted use. ABBV155 (mirzotamab clezutoclax) is the first ADC in this category to reach clinical trials [17, 18].
ADC Linkers
As payload innovation accelerates, linker design has become equally critical to ADC performance. Linkers determine when, where, and how efficiently a payload is released, directly influencing stability, safety, and therapeutic reach. Their behavior in circulation and within the tumor microenvironment often dictates whether even the most potent payloads can achieve meaningful clinical impact.
Cleavable linkers remain the dominant choice in both approved and late-stage ADCs. These chemistries are engineered to respond to specific intracellular or extracellular cues, such as pH changes, redox conditions, or protease activity, ensuring that payloads are released only under the right biological circumstances.
In contrast, non-cleavable linkers offer exceptional stability but rely on complete lysosomal degradation of the antibody to liberate the active drug-linker catabolites. While this reduces off-target toxicity, it also limits membrane permeability and bystander killing, making them less effective in heterogeneous solid tumors [2,19].
This distinction is reflected in the clinical landscape, with only ~18% of FDA-approved ADCs and ~8% of late-stage candidates using non-cleavable linkers, while cleavable Valine-Citrulline (Val-Cit) and Glycine-Glycine-Phenylalanine-Glycine (GGFG) peptide linkers continue to predominate [2].
Their widespread use underscores the field’s reliance on predictable, tumor-activated release mechanisms, yet also highlights the need for next-generation designs that address known limitations.
Linkers implemented in ADCs in late-stage clinical studies
| Linker family | Typical linker examples | Cleavable? | Cleavage mechanism | Class | ADCs Phase 3 |
|---|---|---|---|---|---|
| Val-Cit peptide | mc-Val-Cit-PABC; PEG2-Val-Cit-PABC; Val-Cit-PABA; mc-PEG2-Val-Cit-PABA-Cyc | Yes | Lysosomal cathepsin cleavage of Val-Cit dipeptide; self-immolation | Protease-cleavable | Zilovetramab vedotin; MRG-002; MRG-003; Telisotuzumab vedotin; Sigvotatug vedotin; DP303c; Luveltamab tazevibulin (STRO-002); Trastuzumab duocarmazine (SYD985) |
| GGFG peptide | mc-Gly-Gly-Phe-Gly | Yes | Lysosomal cathepsin cleavage of GGFG tetrapeptide | Protease-cleavable | BNT323/DB-1303; SHR-A1811; Patritumab deruxtecan; Datopotamab deruxtecan; Ifinatamab deruxtecan; Raludotatug deruxtecan |
| β-Glucuronide | Geranyl ketone pyrophosphate oxime ligation (FS-1502) | Yes | β-Glucuronidase-mediated hydrolysis | Enzyme cleavable | FS-1502 |
| Carbonate (CL2A) | Pyrimidine-CL2A-carbonate | Yes | pH-accelerated carbonate hydrolysis | Chemically labile (pH-dependent hydrolysis) | Sacituzumab tirumotecan (MK-2870) |
| Disulfide | SPDB | Yes | Intracellular reduction (glutathione) | Redox-cleavable (non-enzymatic) | Tusamitamab ravtansine (SAR408701) |
| Non-cleavable thioether | Maleimidocaproyl (mc) | No | Payload released after full lysosomal antibody degradation | Proteolytic processing (lysosomal catabolism) | Depatuxizumab mafodotin (ABT-414) |
| Non-cleavable oxime/PEG4 | Hydroxylamine-PEG4 | No | Payload released after antibody catabolism; linker intact | Proteolytic processing (lysosomal catabolism) | ARX-788 |
| Protease-cleavable hydrophilic (proprietary) | Cys-11 (sesutecan) | Yes | Protease-cleavable peptide linker | Protease-cleavable | Rinatabart sesutecan (Rina-S / PRO1184) |
| Undisclosed | Undisclosed | Undisclosed | Undisclosed | Undisclosed | TQB-2102; FDA018; Telisotuzumab adizutecan (ABBV-400); Oportuzumab monatox (Vicinium) |
Information for the table above was partly obtained from Wang et al. (2025). [2]
Emerging ADC linkers
The push to improve linker performance has sparked a wave of innovation aimed at enhancing stability, reducing off-target cleavage, and enabling more precise control over payload release.
Improved Val-Cit linkers
Despite their success, traditional Val-Cit–PAB linkers face challenges, including aggregation, DAR constraints, and susceptibility to premature cleavage by plasma enzymes such as Ces1C and neutrophil elastase [20,21,22]. These vulnerabilities can lead to early payload release and dose-limiting toxicities. To address this, developers have incorporated hydrophilic scaffolds such as PEG or modified peptide sequences (e.g., Glu-Val-Cit, Glu-Gly-Cit) to reduce enzymatic sensitivity and improve stability [20].
More recently, repositioning the Val-Cit motif to the exo-position of the p-aminobenzyl carbamate (PAB) moiety has yielded “exo-linkers” with enhanced hydrophilicity, reduced aggregation, and improved intracellular release profiles [20].
Site-specific linker–payload conjugation
Another major frontier is the shift from heterogeneous to site-specific conjugation. Traditional lysine- or cysteine-based conjugation produces ADCs with variable DAR and inconsistent pharmacokinetics [23,24]. To overcome this, developers are leveraging engineered amino acids, introduced cysteines, or enzyme-mediated conjugation strategies.
Microbial transglutaminase (MTG), for example, enables precise conjugation at Q295 in the Fc region, though this typically requires modification of the nearby N297 glycan. A newer approach pairs MTG with a minimal RKAA peptide linker, enabling direct conjugation to native antibodies and producing homogeneous DAR2 ADCs without altering Fc glycosylation [23]. This represents a meaningful step toward more predictable and manufacturable ADCs.
Tumor-site extracellularly cleaved linkers
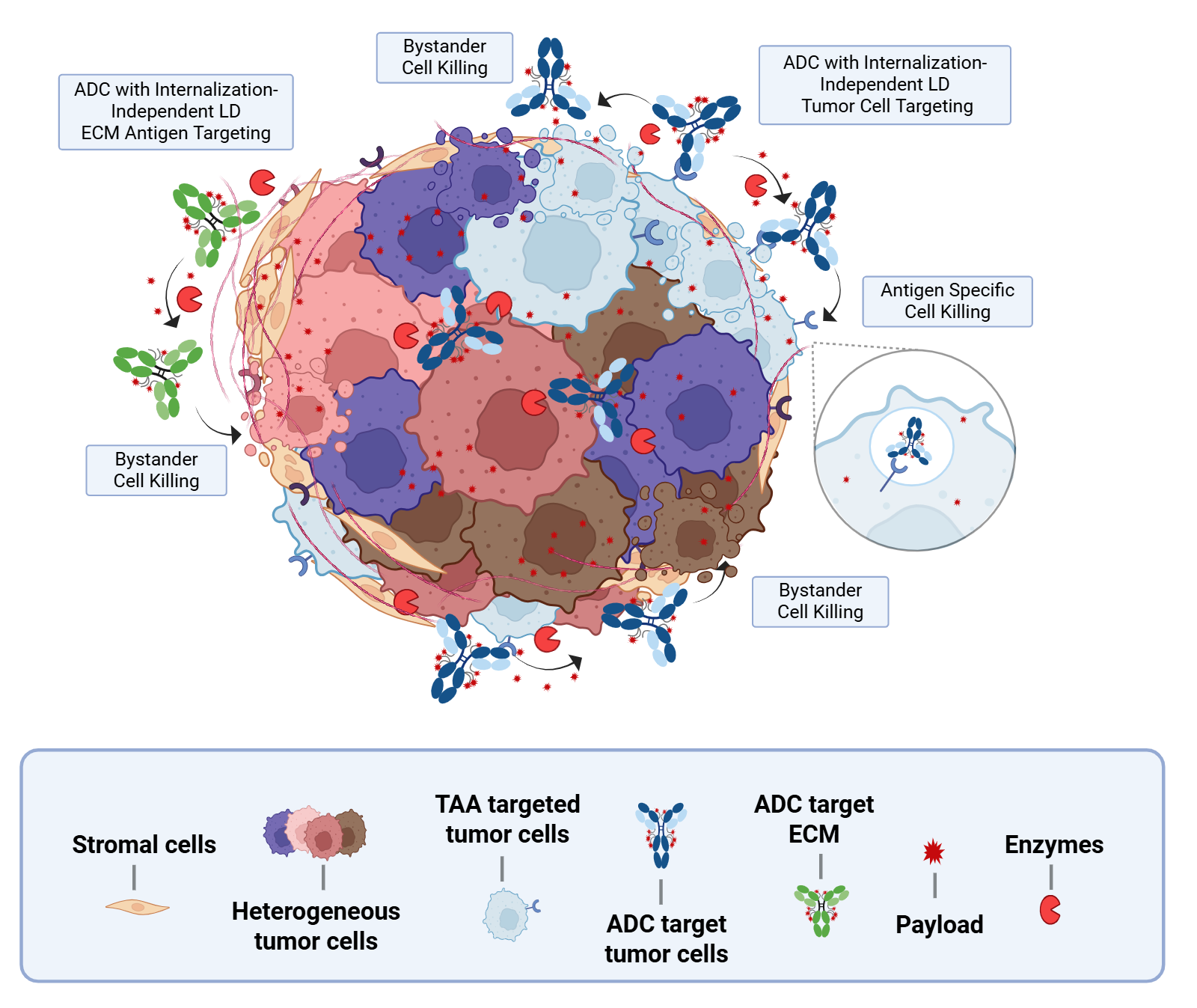
A particularly exciting direction involves linkers designed to be cleaved not inside tumor cells, but within the tumor microenvironment (TME) itself. These linkers exploit enzymes enriched in the TME, such as matrix metalloproteinases, cathepsins, β-glucuronidase, and serine proteases like uPA, to trigger extracellular payload release [25,26,27].
By bypassing the need for internalization, these systems can overcome antigen heterogeneity and enhance bystander killing, broadening the therapeutic reach of ADCs. This strategy is especially promising for solid tumors, where dense stroma and variable antigen expression often limit traditional ADC efficacy.
Nona Biosciences’ proprietary TME-cleavable linker technology exemplifies this emerging class, enabling extracellular payload release driven by tumor-intrinsic enzymes. This approach is being actively advanced in discovery and preclinical programs aimed at expanding ADC utility across challenging solid tumor settings.
Innovative ADC Linker Technology for Extracellular Payload Release
Advancing ADC Development with Nona Biosciences
Nona Biosciences addresses challenges in ADC development through its comprehensive antibody discovery and development platforms based on Harbour Mice®. These platforms excel at identifying and engineering fully human heavy-chain-only HCAb and H2L2 conventional antibodies with exceptional specificity and optimal affinity for tumor-associated antigens, laying a solid foundation for creating effective ADCs.
Reflecting this emphasis on nextgeneration binder discovery, Nona Biosciences has formed a strategic biologics discovery alliance with Valink Therapeutics to accelerate the creation of innovative bispecific antibodies and bispecific ADCs.
Beyond technologies for antibody discovery and development, Nona Biosciences offers end-to-end solutions including conjugation strategies, payload selection, and analytical characterization. This integrated workflow ensures optimal linker-payload design, stability, and manufacturability—key factors in achieving potent anti-tumor activity while ensuring safety.
Related Resources
To explore emerging ADC and site-specific conjugation technologies in greater detail:
- View our Scientific Poster on TME-Cleavable Linker Technology for a detailed look at enzyme-driven extracellular payload release and its application in solid tumor ADC development.
- Read our Case Study on Site-Specific DAR1/DAR2 AOC Conjugation for a technical overview of homogeneous oligonucleotide conjugation strategies using fully human HCAbs.
- PrecisePEG, 2025. [Link]
- Wang R. et al., 2025. [Link]
- Fong A. et al., 2019. [Link]
- Han S. et al., 2022. [Link]
- Morcos A. et al., 2024. [Link]
- Nadal-Serrano M. et al., 2020. [Link]
- Gholami A. et al., 2023. [Link]
- Metrangolo V. et al., 2024. [Link]
- Chen F. et al., 2024. [Link]
- Bukhalid R.A. et al., 2025. [Link]
- ClinicalTrials.gov. [Link]
- Daiichi-Sankyo.us, 2025. [Link]
- ClinicalTrials.gov. [Link]
- Ren P. et al., 2025. [Link]
- ClinicalTrials.gov. [Link]
- ClinicalTrials.gov. [Link]
- Judd A.S. et al., 2024. [Link]
- ClinicalTrials.gov. [Link]
- Choi Y. et al., 2024. [Link]
- Watanabe T. et al., 2024. [Link]
- Dorywalska M. et al., 2016. [Link]
- Zhao H. et al., 2017. [Link]
- Probst P. et al., 2025. [Link]
- Haque M. et al., 2021. [Link]
- Ashman N. et al., 2022. [Link]
- Lopez Rivas P. et al., 2019. [Link]
- Xu K. et al., 2023. [Link]