What are autoimmune diseases?
Autoimmune diseases are a diverse group of chronic conditions prompted by an abnormal immune response against healthy tissues, leading to inflammation and often severe, long-term tissue damage. While the underlying root cause of these heterogeneous conditions remains unclear, the cellular players, namely adaptive immune cells, including B and T cells activated by autoantigens, are known to play a central role in inflammation and tissue damage [1,2].
Globally, autoimmune diseases are estimated to impact 3-5% of the total population and tend to disproportionately affect women over men [1,3]. In the US, the prevalence of autoimmune diseases varies anywhere from ~180–740 per 100,000 individuals for the top ten most common conditions.

Limitations of conventional and targeted therapies in autoimmune diseases
Conventional therapies for autoimmune diseases include small-molecule anti-inflammatory and immunosuppressive drugs that act through different mechanisms, including reducing proinflammatory mediators and inhibiting immune cell activation. These therapies leverage nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and disease-modifying antirheumatic drugs, which are broadly immunosuppressive, can elicit serious adverse effects, and do not address the underlying cause of the disease [1,2].
Targeted therapies, such as small-molecule inhibitors of the JAK-STAT pathway (e.g., ruxolitinib and tofacitinib) and anti-TNFα antibodies (e.g., Adalimumab and Infliximab), enable the specific block of cytokine signaling and activity. Additionally, the adoption of antibody therapeutics targeting CD20, such as Rituximab, originally approved for cancer treatment, enables B-cell depletion and is currently implemented for rheumatoid arthritis, pemphigus vulgaris, and vasculitides [2,4]. However, despite their effectiveness, these therapies are unable to induce full disease remission [1].
The shift toward cellular immunotherapies in autoimmune disease
Similar to the adoption of cancer biologics for autoimmune diseases, CAR-T cells are emerging as an effective strategy for depleting autoreactive B cells and inducing deep, long-lasting remission. While no CAR-T cell therapy targeting B cells has been approved to date for any autoimmune disease indication, a substantial number of clinical trials are underway (i.e., > 80 studies globally) [5,6].
Global Landscape of Autoimmune Disease CAR T Clinical Studies
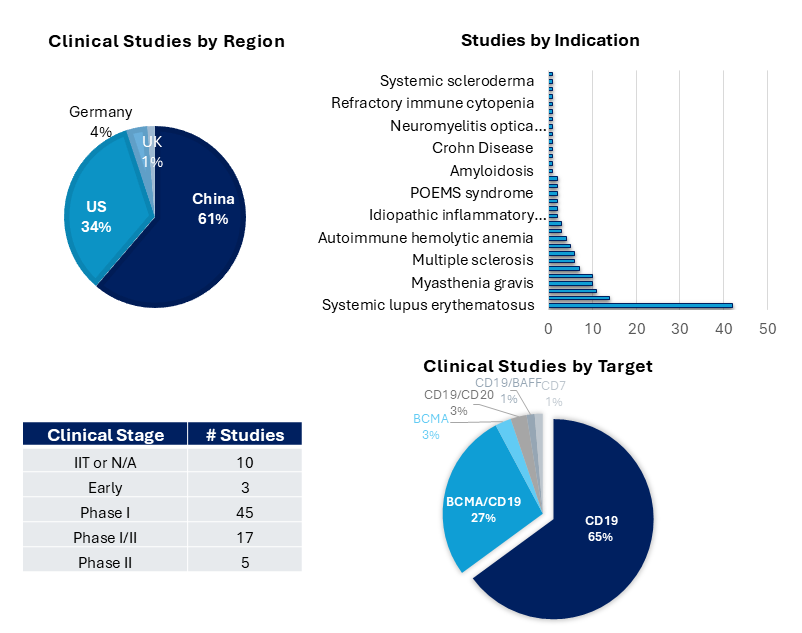
The majority of trials evaluating CAR-T cells in autoimmune diseases are still in early clinical stages and are predominantly conducted in China and the US. These include several Investigator Initiated Trials (IITs) (e.g., NCT06549296, NCT05930314, NCT05859997, NCT05988216, NCT06277427).
IITs provide a versatile clinical mechanism for investigators to explore novel questions, expedite early-stage testing, and generate real-world clinical insights and data to shape future trials.
For CAR-T cell therapies in autoimmune disease, the single biggest advantage of IITs is that they enable rapid, clinician‑driven exploration of immune reset as a completely new therapeutic paradigm, expediting industry timelines, commercial justification, and large‑scale infrastructure. As such, currently, IITs are being leveraged to evaluate safety, tolerability, pharmacokinetics, pharmacodynamics, and preliminary efficacy in various autoimmune disease patient populations.
Among both investigator-initiated and clinical trials, systemic lupus erythematosus, lupus nephritis, ANCA-associated vasculitis, myasthenia gravis, and systemic sclerosis figure prominently as the top five most studied indications.
Not surprisingly, given the success of CD19- and BCMA-targeting CAR-T cells in hematological malignancies, these B-cell receptors are leveraged prominently in autoimmune disease. The majority (~65%) and more advanced studies focus on CD19-targeting therapies because CD19 is broadly expressed across B-cell subsets, from pro-B cells to subsets of plasma cells. Nevertheless, as CD19 expression is downregulated in both short-lived and long-lived plasma cells, a substantial number of studies use combined CD19 and BCMA CAR-T cells to deplete plasma cells.
Relevant B-Cell Receptors for CAR-T Cell Therapies

By depleting populations of autoantibody-producing plasma cells that persist for years, this combined approach can enable long-term remission in autoimmune disease patients [2].
Dual target and combination CAR-T strategies in autoimmune diseases
Currently, BCMA and CD19 are the predominant receptors being targeted for combined CAR-T cell therapies in autoimmune disease, while a minority of studies focus on other combinations, including BCMA/CD20 (NCT06249438), CD19/BAFF (NCT06279923), and CD19/CD20 (NCT06153095).
BCMA and CD19 dual targeting is achieved through different approaches, such as engineered bispecific CARs, co-transduction of T cells with two CARs, or administration of two separate CAR-T cell products, which can be co-infused or sequentially infused.
Combination Strategies for BCMA/CD19 CAR-T Cell Therapies
| Program / ID / Sponsor | Combination Strategy | Setting & phase | Key efficacy signal (high level) |
|---|---|---|---|
| BCMACD19 compound CAR T / NCT05474885 / iCell Gene Therapeutics | Bispecific | Phase 1, open-label | High rate of clinical responses; many patients achieved low disease activity or remission |
| CD19xBCMA dual targeting CAR T / NCT05858684 / RenJi Hospital | Bispecific | Phase 1, dose escalation | Early cohorts show high clinical response rates, with deep B/plasma cell depletion |
| Coinfused CD19 + BCMA CAR T / NCT05030779 / Zhejiang University | Co-infusion | Phase 1, dose-escalation | Deep B cell/plasma cell depletion; frequent drug-free remission/major activity reduction |
| BCMA or CD19 sequential CAR T / NCT06428188 / Essen Biotech | Sequential infusion | Phase 1/2 | Not available |
References for table content: Wang W. et al., 2024; Wu C. et al., 2025; Feng J. et al., 2025 [8–10]
Advancing strategies targeting autoreactive B cells
A caveat of BCMA/CD19 CAR-T cell therapies in autoimmune diseases is the resulting widespread B cell depletion, which weakens immune responses and increases susceptibility to infections. Therefore, emerging trends are leveraging two main approaches to more specifically target autoreactive B cells.
Chimeric Receptor Strategies for T Cell Therapies in Autoimmune Diseases
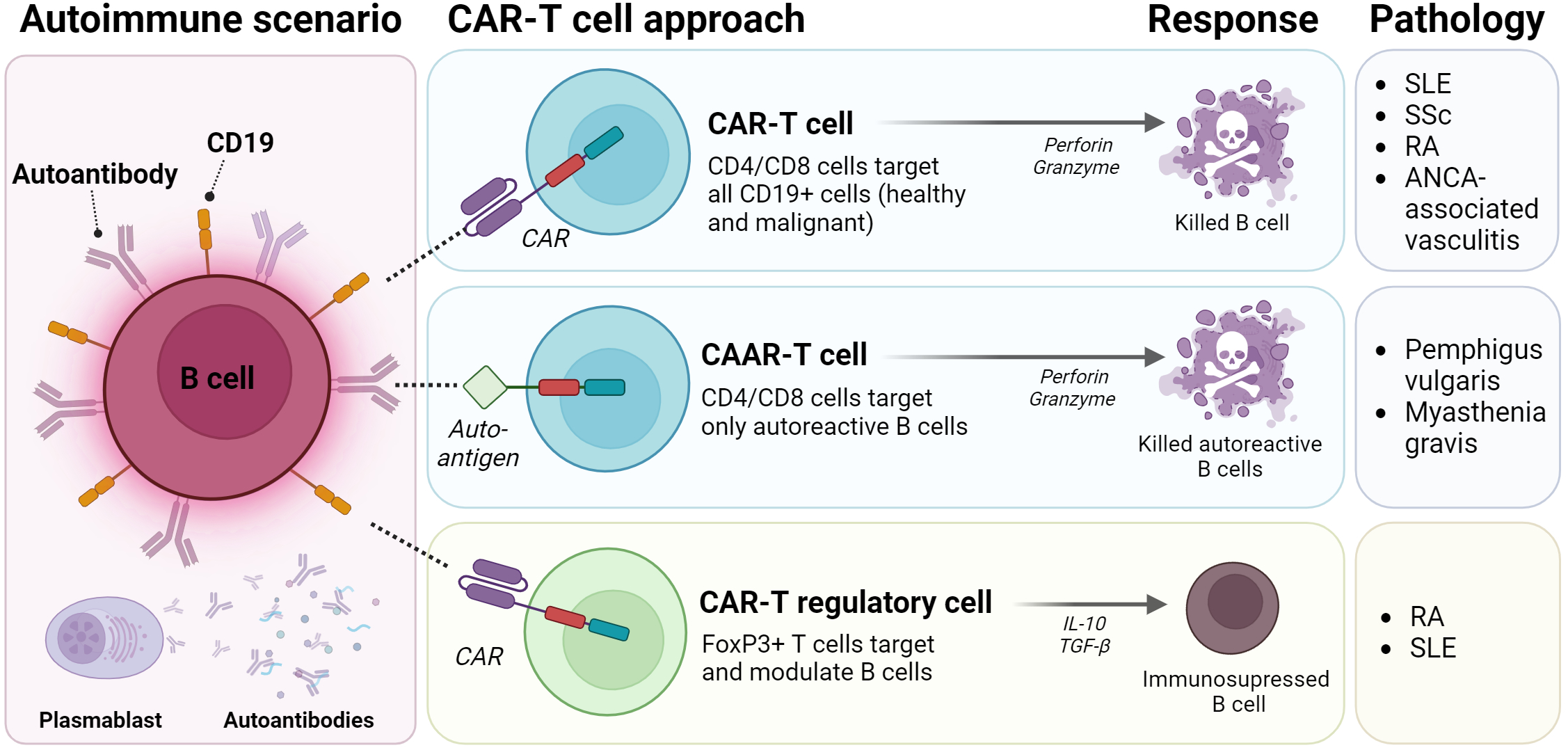
Chimeric Autoantibody Receptor (CAAR) T Cells
– In this modality, the extracellular binding domain of the CAR molecule is replaced with an autoantigen. Binding of autoreactive B cell clones, through B cell receptors (BCRs), to the CAAR in T cells unleashes their cytotoxic activity, leading to specific B cell depletion [1,6,12]. CAAR-T cells were first introduced by Ellebrecht et al. in 2016. The group demonstrated that T cells expressing the autoantigen desmoglein (Dsg) 3 (i.e., Dsg3-CD137-CD3ζ) could kill autoreactive B lymphocytes in vivo in a preclinical model of the antibody-mediated autoimmune disease pemphigus vulgaris (PV) [11]. Currently, CAAR-T cells are being evaluated in clinical Phase 1 and Phase 1/2 trials sponsored by Cabaletta Bio for two indications: muscle-specific tyrosine kinase (MuSK) myasthenia gravis (NCT05451212) and pemphigus vulgaris (NCT04422912).
CAR-T regulatory cells
– This strategy uses T regulatory cells or CD4+ T cells that are modified to overexpress FoxP3 in order to restore immune tolerance. Unlike conventional cytotoxic CAR-T cells, redirected CAR-T regulatory cells exert immunosuppressive functions through various mechanisms, including secretion of anti-inflammatory cytokines (e.g., IL-10 and TGF-β), cytolysis, metabolic disruption, and modulation of antigen-presenting cell maturation or function. Overall, this strategy aims to reduce chronic inflammation associated with autoimmune diseases [1,6,12,13]. Several clinical trials are evaluating CAR-Treg cells in the setting of allogeneic transplantation (cGVHD), including hematopoietic cell transplantation (City of Hope Medical Center- NCT05993611), liver transplantation (Quell Therapeutics Limited- NCT05234190), and renal transplantation (Sangamo Therapeutics- NCT04817774). Significantly, City of Hope Medical Center is also conducting an early-phase 1 study using CD6-CAR-Tregs to target autoreactive effector T cells in patients with type 1 diabetes (NCT07395050). While CAR-Tregs remain at an early stage and challenges remain in their manufacturing and application, they can deliver immune suppression directly to tissues or to antigens driving disease, offering a more precise, tolerance-restoring approach that contrasts with the broad cytotoxic activity of conventional CAR-T cells.
Enabling Next-Generation CAR Therapies for Autoimmune Disease
As CAR-T therapies move into autoimmune indications, success depends on precise immune modulation, durable signaling, and formats that minimize off-target immune suppression. Nona Biosciences’ NonaCAR™ and NonaCARFx™ platforms address these needs by pairing fully human heavy chain-only (HCAb) VH domain binders with functional CAR format screening.
This integrated approach enables the identification of compact, highly expressible binders optimized for CAR signaling, supporting multispecific designs targeting B cells, plasma cells, or autoreactive immune subsets. By evaluating activation and surface expression directly in full CAR constructs, NonaCARFx™ helps de-risk candidate selection and accelerate the development of CAR-T, CAR-NK, CAR-M, and emerging in vivo CAR strategies aimed at achieving deep, durable immune reset in autoimmune disease.
Related Resources
To explore next-generation CAR design and delivery strategies in more detail:
- Download our Poster on Fully Human VH CAR platforms for a concise, visual overview of HCAb-derived VH domains applied in CAR-format screening, functional validation, and targeted delivery workflows
- Read our Blog on Ex Vivo to In Vivo CAR Therapy for insights into how CAR therapies are evolving toward in vivo delivery and expanded therapeutic applications
- Rangel-Peláez C. et al., 2024. [Link]
- Oh S. et al., 2022. [Link]
- Abend A. H. et al., 2024. [Link]
- Damsky W. et al., 2021. [Link]
- Wu D. et al., 2025. [Link]
- Li X. et al., 2025. [Link]
- Robinson W. H. et al., 2024. [Link]
- Wang W. et al., 2024. [Link]
- Wu C. et al., 2025. [Link]
- Feng J. et al., 2025. [Link]
- Ellebrecht C. T. et al., 2016. [Link]
- Cheever A. et al., 2025. [Link]
- Vignali D. A. A. et al., 2008. [Link]